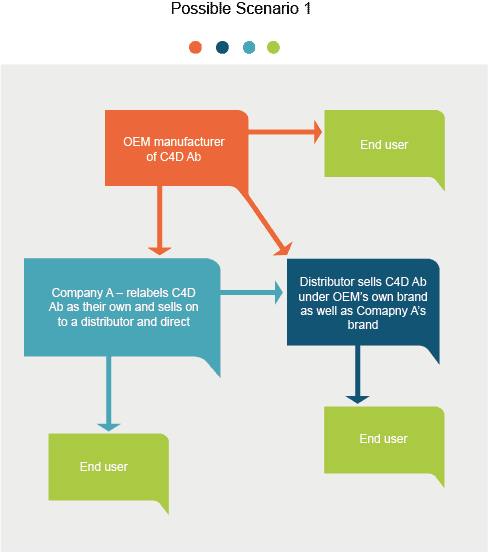
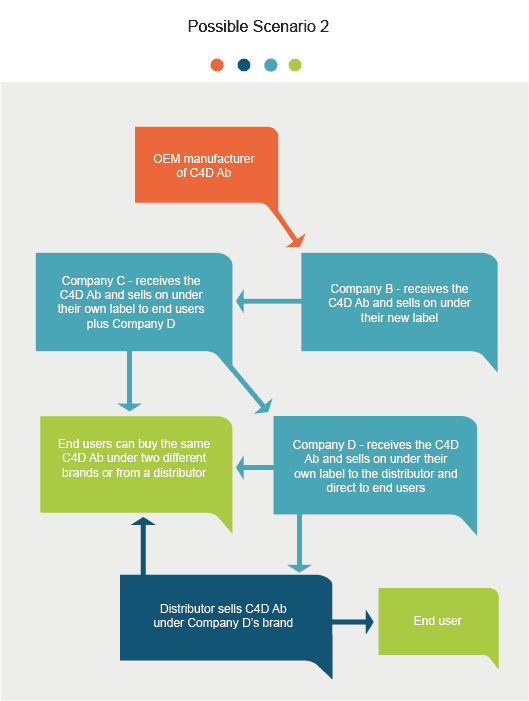
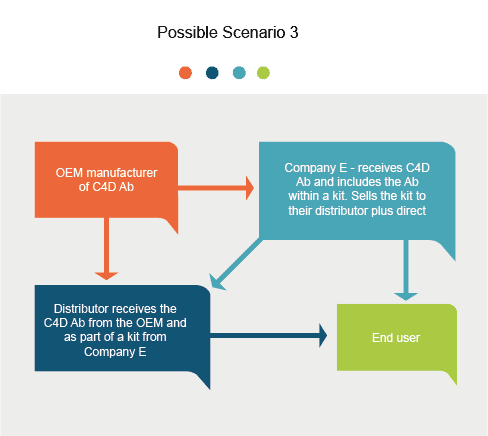
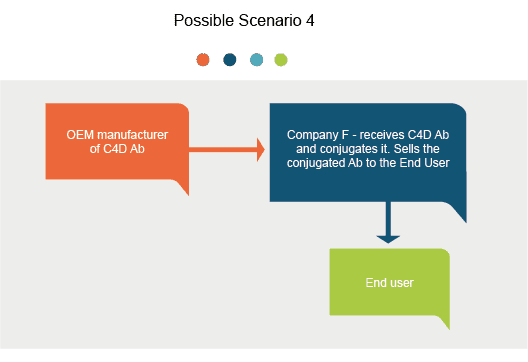
Introduction
The abbreviation “OEM” stands for Original Equipment Manufacturer and the term can be used to describe two processes; ‘OEM in’ and ‘OEM out’. Each industry has its own definition, and within the life science industry they can be defined as follows:
OEM In When you do not manufacture research & IVD reagents but sell another manufacturer’s product under your own label
OEM Out When you are the manufacturer of the research & IVD reagents and sell your product on to a company which markets the products as their own
The question often asked is why OEM?
Frequently a supplier does not have the required framework to produce a large range of highly specialized and high-volume research & IVD reagents themselves, or even if they do, outsourcing the production is simply much cheaper. By employing the services offered by OEMs, they can make use of external production capacities.
OEM solutions are ideal for start-ups which, although adept at design and invention, lack the means or expertise to establish their own factory production. Globalisation has made it much easier for small and medium-sized enterprises (SMEs) to also benefit from this option.
OEM In
Advantages:
Choosing the OEM route generally gives you cost-effective research & IVD reagents – you benefit from the economies of scale which are passed on from the supplier who is able to produce in bulk thereby reducing costs. Outsourcing the job of production can lead to lower prices, higher quality, and more time to focus on your core business.
Summary of benefits:
- Cost effective research & IVD reagents products
- Faster expansion of your existing research & IVD reagents range
- Preserve your own brand
- Maintain your customers
- More time to focus on your core business
Disadvantages:
One of the main hurdles is locating those OEM partners that have that unique life science technology as they are relatively few and far between. Therefore, when it comes to selling your research & IVD reagents products you may find you have no USPs and no exclusivity within the marketplace.
Added to this there is generally very limited scope for your own customisation of the product range.
Lastly, there is no accountability for poor validation or specificity from the OEM and you may unknowingly end up purchasing the same poor-quality antibodies from several different OEM out companies.
Summary of disadvantages:
- Limited novel technology available
- Non-unique products
- Non-exclusivity
- Limited potential to customize products
- Lack of validation
OEM Out
Advantages:
As an OEM out company you generally only deal as a B2B business, and it is a quick way to expand your sales globally by utilising your OEM in clients’ marketing.
Summary of advantages:
- B2B business model
- Quick expansion of sales
- Reduced marketing costs
Disadvantages:
As an OEM out company, the main issue is the negative impact to your own brand. It is important to ensure that the reproducibility, accountability and support you give to your OEM in clients are excellent so they, in turn, can provide a better service to their end users.
Summary of disadvantages:
- Erosion of your own brand
- Limited to B2B sales only